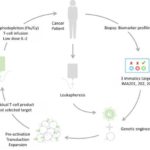
Immatics Announces European Clinical Expansion of its Adoptive Cell Therapy Programs
First patient has been treated in Germany in Immatics’ ACTengine® IMA202-101 trial German regulatory agency, Paul-Ehrlich-Institute (PEI), granted approval to commence another clinical ACTengine® trial in Germany investigating Immatics’ IMA203 product candidate Three clinical trial sites in Germany have started Mehr






